The Preventx Laboratory is a UKAS ISO 15189 accredited medical laboratory, No. 7568.
Where laboratory diagnostic services are provided in whole, or as a part of a product or service offered by Preventx Limited, they are done so in accordance with a Quality System adhering to international standards.
This document summarises key areas of this system that relate to the laboratory services offered to clients.

Facilities
The Preventx Laboratory is located at Meadowhall Business Park, which occupies a prime location on the eastern edge of the Sheffield city centre. The location provides excellent transport and distribution links for sample receipt.
The secure. suite contains both modern office space and a Category 2 wet laboratory across two adjoining sites MBP 4 & 5 for the exclusive use of Preventx.
Assays and Platforms
The Preventx Laboratory is currently accredited to offer the following assays:
- Chlamydia trachomatis (CT) by NAAT PCR test.
- Neisseria gonorrhoeae (NG) by NAAT PCR test.
- Trichomonas vaginalis (TV) by NAAT PCR test.
- Mycoplasma genitalium (MG) by NAAT PCR test.
- HIV-1/2 antibodies and p24 antigen by electrochemiluminescence test.
- Treponema pallidum (SYP) antibodies by electrochemiluminescence test.
- Syphilis TPHA by manual test.
- Syphilis RPR by manual test.
- Hepatitis B surface antigen (HBV – HBsAg) by electrochemiluminescence test.
- Hepatitis B core antibody (HBV – Anti-HBc) by electrochemiluminescence test.
- Hepatitis C antibody (HCV) by electrochemiluminescence test.
- Hepatitis C RNA
The laboratory carries out most testing on the Roche Cobas 6800/8800® and the Roche Cobas e801 systems. The Cobas 6800/8800 provides fully automated sample preparation with real-time polymerase chain reaction (PCR) based NAAT technology for amplification and detection of Chlamydia trachomatis (CT), Neisseria gonorrhoeae (NG), Trichomonas vaginalis (TV), Mycoplasma genitalium (MG). Single (CT) and dual (CTNG) testing services are available. The Cobas e801 platforms provide automated electrochemiluminescence testing for serology tests.
Where other assays are provided by Preventx to support products and services, the samples may be referred to sub-contracted laboratories. Samples referred for testing that include assays other than those above, fall outside the scope of Preventx’s accreditation (and this document) and are offered in accordance with accreditation of the referral laboratory. Samples may be referred to a subcontracted laboratory for confirmatory testing or laboratory continuity (for example, in the case of sustained equipment failure). Such samples will always be referred to a laboratory accredited to similar national or international standards (e.g. ISO15189 or ISO17025).
Where samples are referred to an alternate laboratory, this will be noted on the specimen record and will be fully traceable. For more information about referral laboratories, or details of the testing path for a specific specimen, contact [email protected].
Quality System
Where tested on site in the Preventx Laboratory or referred to an external laboratory, all samples are tested in a laboratory accredited to national or international standards (e.g. ISO15189 or ISO17025).
Preventx follow the relevant national standards (and endeavour to follow future revisions) recommended in UKHSA SMIs, and the ‘Standards for the management of sexually transmitted infections (STIs)’, as published by the British Society for Sexual Health and HIV (BASHH) where possible, with minor variations based on the nature of self-sampling services providing samples of low sample volume.
Preventx completes the NHS Data Security Protection Toolkit annually and is registered with the ICO. Patient information is collected, stored and processed securely and consent to process data is obtained in line with GDPR requirements and Cyber Essentials plus.
Sample Handling
All samples received into the laboratory are stabilised and prepared for testing as soon as possible after receipt (usually within 5 hours). In instances where timely stabilisation is not possible, and where appropriate, samples are refrigerated (between 2-8°C) until preparation can be carried out.
A study carried out by Preventx has shown that samples transported though the Royal Mail and remaining un-refrigerated up to 30 days (CT/NG), 12 days (HIV/SYP/Anti-HBc/HCV), 8 days (HBsAg) still produce accurate test results. Any samples the laboratory believe to have not been refrigerated or stabilised within these periods from collection are still tested (as positive results would likely still be detected) however negative outcomes are handled as ‘out-of-validation’ as it is believed that the negative result has a slightly increased probability of being false. There are, however, very few samples to which this outcome is applied (only a small number of samples during heavy postal periods, such as the winter festive season).
While stability of detectable HIV & SYP antibodies in blood samples is likely to be beyond that quoted, the limiting factor is the haemolysation of blood which occurs naturally in transported samples and becomes the barrier to testing. As a result, the haemolysis level of every sample is quantified prior to analysis during routine quality checks.
Once appropriately prepared, all samples are handled, tested and stored according to manufacturer guidelines or otherwise validated processes. Samples are then queued to be tested in-house or referred to be tested at an alternative laboratory. Preventx endeavour to test at least 99% of samples within 5 working days of receipt regardless of result; however, most samples are tested within 48 hours.
Sample and Form Retention
Where available, neat primary samples received by Preventx are retained until a result is submitted for the sample. Stabilised samples for all tests are retained for a minimum of 7 days, during which they may be re-tested by request at the Preventx Laboratory or an external laboratory if a third-party confirmation is required.
Forms received with samples are retained for a minimum of 2 weeks, and in some cases may be stored electronically and attached to the samples specimen record in the Preventx Specimen Management system.
Result Delivery
All test results are stored by Preventx in a secure central database however the result delivery channels may vary based on client agreements. In most cases, results are available via the Preventx Specimen Management system.
While the resulting outcome and associated data will always be clearly available, the web-based result and sample management systems are continually under development and improvement, so the presentation of results may evolve over time. Presentation of results may also vary dependent on the service being used (for example sh.uk vs freetest.me)
Nationally Reported Datasets
The Preventx Laboratory submits the mandatory CTAD reporting data to UKHSA on a quarterly basis.
Preventx also submit other public health datasets as required (e.g. GUMCAD for appropriate services).
Testing
Test Accuracy – 6800/8800
Platform performance characteristics as stated and validated by manufacturer:
| Sample Type | Performance Characteristic | Assay Target | |||
|---|---|---|---|---|---|
| Chlamydia trachomatis (CT) | Neisseria gonorrhoeae (NG) | Trichomonas vaginalis (TV) | Mycoplasma genitalium (MG) | ||
| Urine (male) | Sensitivity (%) | 100 | 100 | 100 | 100 |
| Specificity (%) | 99.7 | 99.5 | 99.3 | 98.7 | |
| CV (%) | 2.94 – 4.05 | 1.17 – 3.55 | 1.5 – 2.4 | 1.2 – 3.3 | |
| Urine (female) | Sensitivity (%) | 95.6 | 94.8 | 100 | 100 |
| Specificity (%) | 99.7 | 99.9 | 99.7 | 97.7 | |
| CV (%) | 2.94 – 4.05 | 1.17 – 3.55 | 1.5 – 2.4 | 1.2 – 3.3 | |
| Vaginal swab | Sensitivity (%) | 99.2 | 100 | 100 | 90.9 |
| Specificity (%) | 99.0 | 99.7 | 99.4 | 99.4 | |
| CV (%) | 1.62 – 3.32 | 2.04 – 3.34 | 2.4 – 2.8 | 2.3 – 4.3 | |
| Rectal swab | Sensitivity (%) | 95.1 | 99.0 | N/A | N/A |
| Specificity (%) | 99.2 | 99.3 | N/A | N/A | |
| CV (%) | 1.62 – 3.32 | 2.04 – 3.34 | N/A | N/A | |
| Pharyngeal swab | Sensitivity (%) | 100 | 100 | N/A | N/A |
| Specificity (%) | 99.8 | 98.8 | N/A | N/A | |
| CV (%) | 1.62 – 3.32 | 2.04 – 3.34 | N/A | N/A | |
The platforms Positive Predictive Value (PPV) and Negative Predictive Value (NPV) can be calculated based on the prevalence among patients testing. While Preventx monitor PPV and NPV using live prevalence data, the following table shows the hypothetical PPV and NPV attained for a given prevalence (and based on Roche’s overall sensitivity and specificity values).
| Prevalence (%) | Chlamydia trachomatis (CT) | Neisseria gonorrhoea (NG) | ||
| PPV (%) | NPV (%) | PPV (%) | NPV (%) | |
| 1 | 63.89 | 99.95 | 86.86 | 99.96 |
| 3 | 84.41 | 99.86 | 95.29 | 99.89 |
| 5 | 90.21 | 99.77 | 97.18 | 99.81 |
| 10 | 95.11 | 99.51 | 98.64 | 99.61 |
| 15 | 96.87 | 99.22 | 99.14 | 99.38 |
| 20 | 97.77 | 98.89 | 99.39 | 99.12 |
| 30 | 98.69 | 98.12 | 99.64 | 98.50 |
| 50 | 99.43 | 95.72 | 99.85 | 96.58 |
Where prevalence drops and the estimated PPV falls below 90%, confirmatory testing will be carried out on all positive samples using an alternative NAAT target. In accordance with UKHSA guidelines, confirmatory testing will be performed on existing material to negate the need for patient contact and further sampling.
Test Accuracy – E801
Platform accuracy as stated and validated by manufacturer:
| Assay | Clinical Sensitivity (%) | Clinical Specificity (%) |
|---|---|---|
| Elecsys HIV Duo (HIV) | 100% | 99.63% |
| Elecsys Syphilis (SYP) | 100% | 99.88% |
| Elecsys Anti-HCV II (HCV) | 100% | 99.66% |
| Elecsys Anti-HBc II (Anti-HBc) | 100% | 99.60% |
| Elecsys HBsAg II (HBsAg) | 100% | 99.88% |
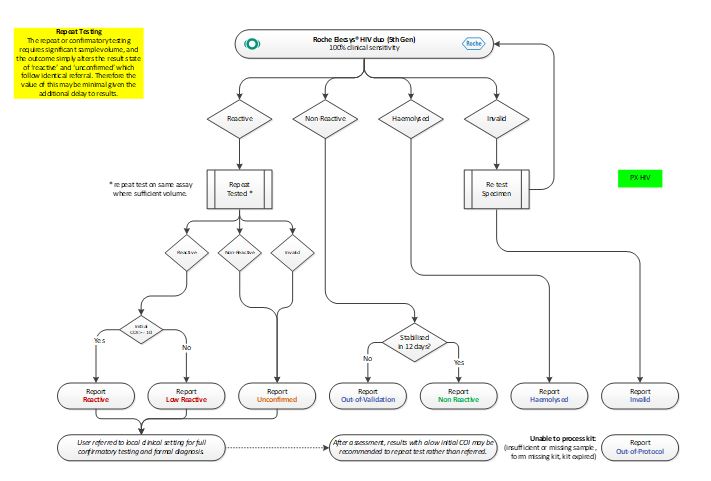
The HIV Duo test provides independent sub-results for HIV-1/2 antibodies, and HIV p24 antigens simultaneously, alongside a calculated overall result.
Recommended window period:
| Assay | Weeks to achieve max sensitivity |
|---|---|
| Elecsys HIV Duo (HIV) | 4-8 |
| Elecsys Syphilis (SYP) | 6-8 |
| Elecsys Anti-HCV II (HCV) | 4-10 |
| Elecsys Anti-HBc II (Anti-HBc) | From onset of symptoms |
| Elecsys HBsAg II (HBsAg) | 4-9 |
In most test settings and sampling kits, Preventx quote:
“the test is highly accurate after 4 weeks and almost 100% after 8 weeks“
Testing Algorithm – CT / NG / TV / MG
The testing algorithm used for testing samples is based on the PHE’s UK Standards for Microbiology Investigations and BASHH guidelines with adjustments for dual (CT & NG) & (TV & MG) testing and platform manufacturer test methods.
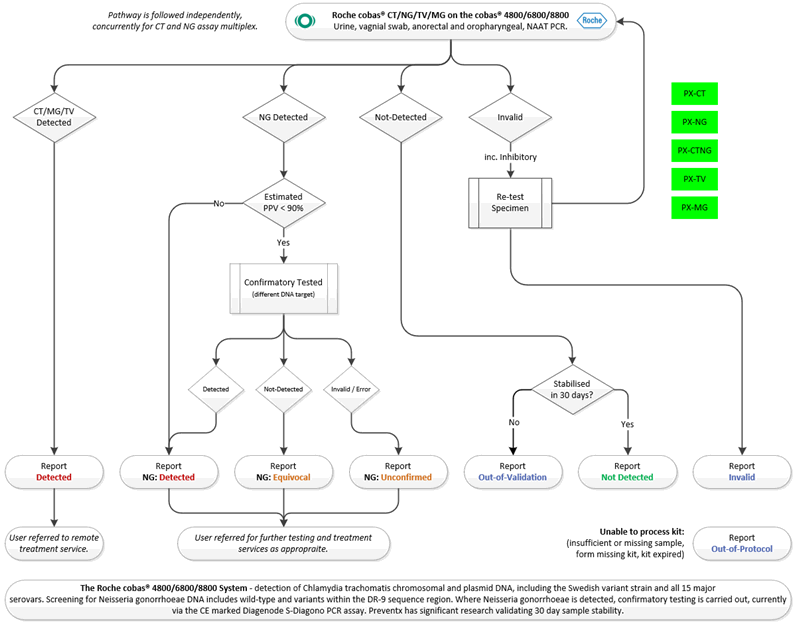
Where samples are unable to be tested (for example, there was no sample provided, or the kit was not completed correctly) an ‘out of protocol’ result will be submitted. In cases where not enough sample is present for repeat testing, the ‘Invalid’ test result outcome will be followed and an ‘Equivocal’ result will be submitted.
Where CT and NG are both detected, multiple test flow routes are followed (i.e. ensuring each submitted result has completed its respective process as per above). Estimated PPV values are evaluated based on regular analysis of overall testing positivity.
Testing Algorithm – HIV
The testing algorithm used for testing samples is based on the PHE’s UK Standards for Microbiology Investigations Anti-HIV screening and BASHH guidelines with adjustments for platform manufacturer test methods and internal testing specification.
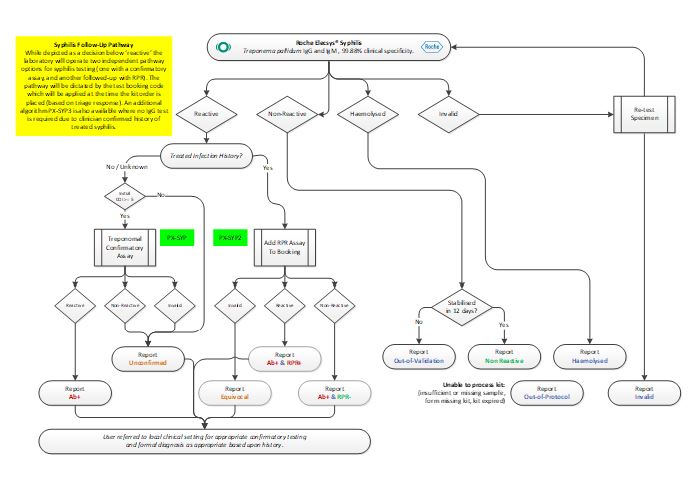
Testing Algorithm – SYP
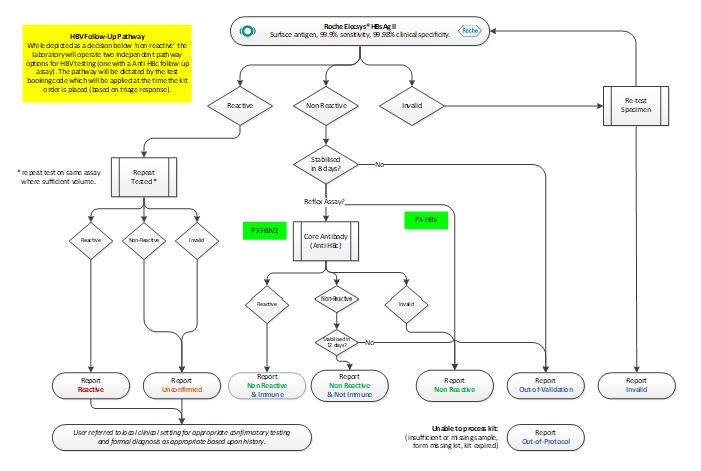
Testing Algorithm – HBV
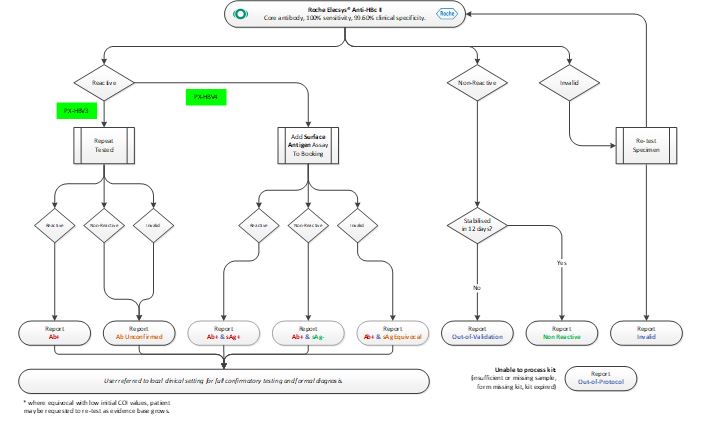
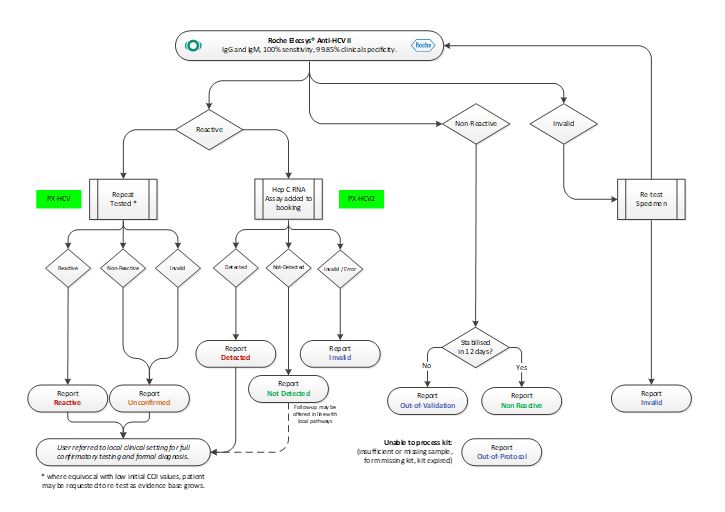
Testing Algorithm – HCV
Services and Variations
While individual agreements with clients may apply variations to the services offered (and take precedence over the service specification outlined in this document), the following Preventx services include accredited testing:
- Freetest.me (www.freetest.me)
Online (and offline) self-sampling kits for STI testing, offered free of charge to users and funded by the users local NHS Trust or Local Authority where available. Results and onward clinical management for patients using freetest.me services are handled via local service provides (usually the local NHS trust) or Preventx’s partner (e.g. THT).
- Sexual Health London (www.shl.uk)
Often referred to as the pan-London e-Service, SHL offers a free STI testing and treatment across most London boroughs. The service operates under clinical oversight from Chelsea and Westminster Hospital NHS Foundation Trust who also provide patient management services, with postal treatment provided by Lloyds Pharmacy Online Doctor.
- SH.UK (www.sh.uk)
Preventx’s new SH service encompasses all types of service. The service operates in a similar way to the services listed above, but provides one central patient experience for online/postal testing. Patient management and onward clinical care of patients is managed via local service and partners.
Where a service has been commissioned, Preventx’s clinical governance requires that a satisfactory care pathway is in place for service users to ensure that result interpretation and onward management and/or treatment services can be provided (directly or via referral).
Responsibilities
Preventx will be responsible for:
- Offering self-sampling testing services to patients who meet eligibility requirements agreed with the client (e.g. geographic region and age) and offline SmartKit testing to all patients who have completed returned test kits (unless excluded).
- Testing samples as per the agreement, and providing results to the provider or organisation responsible for patient care.
- Sending notifications directly to patients; however, where such messages include test result interpretations, the client or provider assumes responsibility for the content of such messages (agreed by acceptance of this document).
The client or local provider, as appropriate, will be responsible for:
- Continuation of the patient care pathway following test results being released, including provision of treatment.
- The interpretation and notification of results to patients (may be automated by Preventx on the customers behalf).
- Handling patient enquiries, in particular those of a clinical nature (for example, queries about results or treatment options).
- Managing existing or future medical issues relating to the patients testing or treatment outcomes.
Queries and Complains
Queries and complaints may be submitted via postal correspondence or email to: [email protected]
The Preventx Laboratory will follow a documented complains procedure where a complaint is submitted.
